
Should megaprosthesis implants be a viable option in elderly patients after distal femur and periprosthetic distal femur fractures?—a retrospective cohort study
Highlight box
Key findings
• Megaprosthesis may be a reliable treatment in elderly patients with distal femur fractures or periprosthetic distal femur fractures with good patient-reported outcomes at the minimum follow-up of 12 months.
What is known and what is new?
• The treatment of distal femur fractures and periprosthetic distal femur fractures in elderly patients represents a complex challenge for knee surgeons. Not all fractures may be treated with retrograde intramedullary nailing or plate because of comminution, poor bone quality or implant loosening. This study aims to analyze the clinical outcomes and complications of patients undergoing megaprosthesis replacement in the management of these fractures.
What is the implication, and what should change now?
• Megaprosthesis implants could provide a rapid return to daily activities with immediate full weight-bearing, range of movement recovery, and joint function preservation in elderly patients. However, complications could be challenging to manage.
Introduction
Distal femur fractures (DFFs) account for 3–6% of adult femoral fractures and 0.3% of overall fractures. DFFs are characterized by a bimodal distribution with a higher incidence among young men following high-energy injuries and older women after low-energy accidents. Risk factors such as osteoporosis, obesity, and neurologic disorders are common in elderly patients (1-4). Periprosthetic distal femur fractures (PDFFs) represent a consistent part of DFFs and are expected to grow dramatically in the following years due to the rising number of primary total knee arthroplasties (TKA) and the aging population (1). PDFFs are the most frequent fracture around knee prosthesis, with a reported incidence between 0.3–2.5% of primary TKAs, followed by tibial and patellar periprosthetic fractures (1,2). Older women are more typically involved in these fractures, and several risk factors, such as osteoporosis, obesity, rheumatoid arthritis, chronic steroid use, diabetes mellitus, and neurologic disorders, have been described (1-3). Surgical-related risk factors were also reported, including anterior femoral notching, component malalignment, and rotationally constrained implants (5,6).
The primary treatment for DFFs and PDFFs is open reduction and internal fixation (ORIF) with a locking plate or retrograde intramedullary nailing (RIMN) with different indications (7,8). RIMN provides a less invasive surgical procedure, while ORIF improves fixation in osteoporotic bone (9,10). In contrast, RIMN and ORIF are not always feasible: RIMN is not suitable in comminuted articular fractures and closed box prosthetic components configurations, while both RIMN and ORIF are not available in cases of severe bone loss, poor bone quality, or periprosthetic fractures with femoral component loosening (9,11,12). Moreover, ORIF and RIMN in treating DFFs and PDFFs can lead to long-lasting, technically challenging surgeries and long-term postoperative recovery with prolonged bed rest and no weight-bearing (8). Due to these limitations, knee prosthetic replacement with distal femur replacement (DFR) implant to manage bone loss could be a viable solution for some DFFs and PDFFs, providing a rapid return to daily activities with immediate full weight-bearing, full range of motion (ROM), and joint function preservation (11,13,14).
The appropriate treatment, especially in elderly patients with symptomatic knee osteoarthritis, multi-fragmentary fractures, poor bone quality, or femoral prosthetic loosening, is still under debate (7-15). This study aims to analyze patient-reported outcomes measures (PROMs) and intra- and postoperative complications of elderly patients undergoing megaprosthesis implants for DFFs or PDFFs. We present this article in accordance with the STROBE reporting checklist (available at https://aoj.amegroups.com/article/view/10.21037/aoj-23-21/rc).
Methods
A retrospective cohort study was carried out on a consecutive series of 20 patients treated with DFR at the Orthopaedic and Traumatology Department at Orthopaedic and Trauma Center (CTO) Hospital of Turin between January 1st, 2017 and January 1st, 2021.
The Institutional Review Board (IRB) of the CTO Hospital of Turin defined this study as exempt from IRB approval (retrospective study on a well-established surgical procedure) and conducted in accordance with the Declaration of Helsinki (as revised in 2013). All patients were informed about the study and consented to participate.
Inclusion and exclusion criteria
Inclusion criteria were age over 65 years, diagnosis of DFF or PDFF treated with DFR implant, and a minimum of 12-month follow-up (FU) after surgery.
Study exclusion criteria were previous prosthetic infection, aseptic tibial or femoral component loosening, failure of previous distal femur treatment with ORIF or RIMN, polytrauma patients, and patients diagnosed with the oncologic disease. Two different authors (D’Antonio, F Bosco) evaluated each eligible case independently. A third author (F Giustra) was involved in solving any doubts.
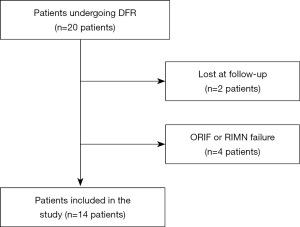
Twenty patients underwent DFR during the study period. Two patients were lost at the final FU; three were excluded because of previous ORIF failure, and one was due to RIMN failure. Fourteen consecutive patients were thus selected in the analysis of this study according to the inclusion and exclusion criteria. The patients’ inclusion procedure is summarized in Figure 1.
Preoperative assessment
At the time of the study, the indication for DFR after DFF or PDFF was based on clinical and radiographic parameters. Elderly patients with DFF and a history of knee osteoarthritis, multi-fragmentary fracture, poor bone quality, or PDFF with severe bone loss or femoral component loosening were eligible for DFR surgery. The patient’s fractures were evaluated with standard anteroposterior (AP) and lateral X-rays and a targeted computed tomography (CT) scan of the involved body segment. Moreover, three-dimensional (3D) CT scans are routinely developed at our institution. Hence, each patient received personalized preoperative planning based on CT scans to correctly determine the resection height, megaprosthesis design, and modular characteristics. The Arbeitsgemeinschaft für Osteosynthesefragen/Orthopaedic Trauma Association (AO/OTA) classification was applied to analyze DFFs. Rorabeck and Taylor’s classification, Su’s, and Kim’s classification were all used to analyze PDFFs (16-18). Rorabeck and Taylor described three types of periprosthetic distal femoral fractures that require different treatments according to component stability, fracture displacement, and comminution (16). Su et al. described an operative classification for PDFF according to the height of the fracture relative to the femoral component (17). Kim’s classification was recently developed to analyze bone stock quality, prosthesis fixation, and reducibility of the fracture (18). Patients with DFF were also assessed for osteoarthritis history and according to the Kellgren-Lawrence (KL) scale before surgery.
Surgical technique
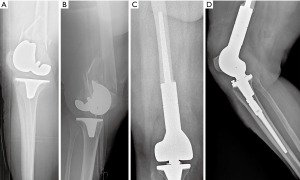
Careful preoperative planning is performed before the surgery based on CT scans, AP, and lateral X-rays. It is crucial to properly establish the transection height as distal as possible above the fracture. The surgical access is a median skin incision and a medial parapatellar arthrotomy obtaining extensive exposure, sometimes requiring a tibial tuberosity osteotomy to preserve the extensor mechanism from iatrogenic injuries. After identifying the fracture, if a femoral component is present, it is removed in combination or separately with the fractured area, and subsequently, the tibial component is removed. Tibial preparation is then performed according to the surgical technique. Transection of the distal femur is performed after marking the correct height on the distal femur. The new cemented prosthesis is implanted following the specific surgical procedure. Three different implant types were used for DFR during the study period: the Zimmer Orthopedic Salvage System (OSS®), the Zimmer Segmental System (ZSS®), or the Megasystem-C® Link, depending on hospital availability. Pre- and post-operative images of DFF and PDFF treatment with megaprosthesis implant are reported in Figures 2,3.

Postoperative program
After surgery, early knee mobilization exercises for full ROM recovery, quadriceps strengthening, and gait rehabilitation were performed from the first postoperative day under the supervision of a physiotherapist. Early full weight-bearing with crutches or walkers was allowed for 4 weeks. At the first-month clinical and radiographic FU, progressive removal of walking aids was permitted.
Data extraction
Data were obtained from departmental databases, FU examinations, and telephone interviews. All the patients involved in the study were investigated for the following demographic characteristics and clinical parameters: trauma energy, age at the time of surgery, gender, body mass index (BMI) and comorbidities, operative time, length of hospital stay (LOS), time to total weight bearing. Blood loss was assessed by evaluating the need for transfusions; intra and postoperative complications and death after surgery were also reported. Patients were examined one, three, and six months after surgery and followed up with clinical and radiographic examinations every six months for a minimum of 12 months. At the final FU, postoperative ROM, the Knee Society Score (KSS) knee score, KSS function score, and the Oxford Knee Score (OKS) were assessed for each patient (19,20).
Statistical analysis
Statistical analysis was performed using the Statistical Software MedCalc (MedCalc Software, Ostend, Belgium). All data were analyzed with descriptive statistics developing mean and median values and standard deviation for continuous variables. Absolute frequencies and percentages were calculated for categorical variables. Microsoft Excel spreadsheets (Version 2019, Microsoft, Redmond, WA, USA) were used to collect data.
Results
Demographic data
The main demographic characteristics and comorbidities are shown in Table 1. Thirteen women and one man were included in the study. Five patients were diagnosed with DFF, and nine were diagnosed with PDFF. The mean age at intervention was 82.1±7.6 years. The median age value at intervention was 84 (range, 71–93) years. The mean BMI was 28.6±7.3. The median value of BMI was 26.6 (range, 20.8–43.0). The mean FU period was 30.7±18.1 months. The median value of FU was 26.5 (range, 69.0–12.0) months.
Table 1
| Cases | Age (years) | Sex | BMI (kg/m2) | Comorbidities |
|---|---|---|---|---|
| 1 | 71 | F | 43.0 | Hypertension, allergic asthma |
| 2 | 84 | F | 43.0 | MADD, UTI, spinal stenosis |
| 3 | 89 | F | 21.5 | Hypertension |
| 4 | 72 | F | 30.9 | * |
| 5 | 87 | F | 23.0 | MADD, hypertension |
| 6 | 72 | F | 31.3 | Hypertension, multiple sclerosis |
| 7 | 84 | F | 20.8 | * |
| 8 | 93 | F | 26.8 | Chronic gastritis, venous insufficiency, osteoporosis |
| 9 | 84 | F | 23.4 | Dyslipidemia |
| 10 | 85 | M | 26.4 | Diabetes mellitus, hypertension, pacemaker wearer |
| 11 | 88 | F | 22.9 | Hypertension, Arrhythmia |
| 12 | 75 | F | 30.5 | Hypertension, PHR, bilateral glaucoma |
| 13 | 75 | F | 23.3 | Bilateral TKA, hypertension, MADD |
| 14 | 91 | F | 33.9 | MADD, hypertension, COPD, breast cancer, NHL, left THA |
*, no comorbidities. F, female; M, male; BMI, body mass index; MADD, mixed anxiety depressive disorder; UTI, urinary tract infections; PHR, partial hip replacement; TKA, total knee arthroplasty; COPD, chronic obstructive pulmonary disease; NHL, non-Hodgkin lymphoma; THA, total hip arthroplasty.
Fractures’ characteristics
The fractures’ characteristics and classification are described in Table 2. The injury was related to low-energy trauma in all patients. Concerning the implant types, the choice was consequent to hospital availability. Only two patients with PDFF had previously undergone surgery at our institution; the other seven received primary TKA in a different hospital. The average time between the first implantation and DFR surgery was 7.2±4.4 years.
Table 2
| Cases | Type of fracture | Fracture classification | Surgical duration (minutes) | Length of stay (days) | Transfusions | Implant characteristics | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Kellgren-Lawrence | AO/OTA | Rorabeck and Taylor | Su | Kim | Femur | Tibia | |||||
| 1 | PDFF | NA | NA | 3 | 3 | 3 | 195 | 20 | X | ZSS (Zimmer) | RHK (Zimmer) |
| 2 | PDFF | NA | NA | 2 | 3 | 2 | 240 | 63 | – | ZSS (Zimmer) | RHK (Zimmer) |
| 3 | PDFF | NA | NA | 3 | 2 | 2 | 135 | 10 | X | ZSS (Zimmer) | RHK (Zimmer) |
| 4 | DFF | III | 33C2 | NA | NA | NA | 155 | 9 | – | Megasystem-C (Link) | Megasystem-C (Link) |
| 5 | PDFF | NA | NA | 3 | 3 | 3 | 145 | 14 | – | OSS (Zimmer) | OSS (Zimmer) |
| 6 | PDFF | NA | NA | 3 | 3 | 3 | 160 | 14 | – | Megasystem-C (Link) | Megasystem-C (Link) |
| 7 | PDFF | NA | NA | 3 | 3 | 3 | 155 | 17 | – | OSS (Zimmer) | OSS (Zimmer) |
| 8 | PDFF | NA | NA | 3 | 2 | 3 | 120 | 15 | – | OSS (Zimmer) | OSS (Zimmer) |
| 9 | DFF | III | 33C2 | NA | NA | NA | 140 | 12 | – | OSS (Zimmer) | OSS (Zimmer) |
| 10 | PDFF | NA | NA | 3 | 3 | 3 | 120 | 14 | – | ZSS (Zimmer) | RHK (Zimmer) |
| 11 | DFF | IV | 33C3 | NA | NA | NA | 205 | 25 | X | OSS (Zimmer) | OSS (Zimmer) |
| 12 | DFF | III | 33C3 | NA | NA | NA | 140 | 20 | X | OSS (Zimmer) | OSS (Zimmer) |
| 13 | PDFF | NA | NA | 3 | 3 | 3 | 140 | 17 | – | OSS (Zimmer) | OSS (Zimmer) |
| 14 | DFF | III | 33C3 | NA | NA | NA | 140 | 29 | – | OSS (Zimmer) | OSS (Zimmer) |
AO, Arbeitsgemeinschaft für Osteosynthesefragen; OTA, Orthopaedic Trauma Association; PDFF, periprosthetic distal femur fracture; NA, not applicable; ZSS, Zimmer Segmental System; RHK, rotating hinge knee; DFF, distal femur fracture; OSS, Orthopedic Salvage System; X, transfusion performed; –, no transfusion.
Clinical data
The mean operative time was 156.4±34.2 minutes. The median value of operative time was 142.5 (range, 120–240) minutes. The mean hospital stay was 19.9±13.6 days. The median value of hospital stay was 16 (range, 9–63) days. Four patients received red blood cell (RBC) transfusions after surgery. All patients effectively underwent the previously described rehabilitation program and achieved full weight-bearing between the first and third postoperative month.
PROMs
All PROMs are reported in Table 3. At the final FU, the mean KSS knee score was 79.5±11.2; the mean KSS function score was 69.0±17.9. The median value of the KSS knee was 82 (range, 58–94); the median value of the KSS function was 70 (range, 40–91). The mean OKS score was 31.6±8.9. The median value of the OKS score was 33.5 (range, 15–42). The mean ROM was 99.3°±9.8°. The median value of ROM was 100° (range, 80°–120°).
Table 3
| Cases | PROMs | Postoperative ROM (degrees) | Complications | Follow-up duration (months) | ||
|---|---|---|---|---|---|---|
| KSS knee | KSS function | OKS | ||||
| 1 | 74 | 65 | 25 | 95 | Anemia, prosthetic infection, implant loosening | 69 |
| 2 | 64 | 45 | 16 | 95 | Delayed wound healing | 59 |
| 3 | 94 | 90 | 42 | 120 | Anemia, delayed wound healing | 49 |
| 4 | 90 | 85 | 36 | 105 | – | 38 |
| 5 | 89 | 90 | 40 | 105 | – | 37 |
| 6 | 87 | 80 | 39 | 100 | – | 35 |
| 7 | 85 | 80 | 41 | 95 | – | 29 |
| 8 | 92 | 91 | 38 | 100 | – | 24 |
| 9 | 85 | 75 | 35 | 105 | – | 19 |
| 10 | 73 | 65 | 31 | 90 | – | 18 |
| 11 | 76 | 60 | 30 | 110 | Anemia | 15 |
| 12 | 67 | 50 | 23 | 100 | Anemia | 14 |
| 13 | 79 | 50 | 32 | 90 | – | 12 |
| 14 | 58 | 40 | 15 | 80 | – | 12 |
PROMs, patient-reported outcome measures; KSS, Knee Society Score; OKS, Oxford Knee Score; ROM, range of motion; –, no complication.
Complications
The reported complications are shown in Table 3. The overall complication rate was 35.7% (five patients), with a re-intervention rate of 21.4% (three patients) and implant removal of 7.1% (one patient). Two delayed wound healings requiring surgical wound debridement were recorded. One prosthetic joint infection (PJI) case with chronic osteomyelitis and implant mobilization was reported. There were no intraoperative fractures or extensor mechanism injuries. No peroneal nerve palsy was reported. No deaths were observed at the last FU.
Discussion
The main result of this study is that DFR after DFF or PDFF seems to be a suitable option in elderly patients with comminuted multi-fragmentary fractures, prosthetic implant loosening and lack of residual bone stock with good clinical outcomes and an early return to ambulation.
Regarding demographic characteristics, this study reveals a higher incidence of DFFs or PDFFs in older women with comorbidities such as osteoporosis, obesity, rheumatic disease, diabetes mellitus, and neurologic disorders, as described in the literature (21-25).
The mean operative for DFR after DFF or PDFF reported in the literature varies widely. Jassim et al., in their case series, described a mean operative time of 140 minutes (24), Matar et al. reported a mean operative time of 128 minutes (22), while Sukhontamarn et al. showed a mean operative time of 125 minutes (25). In this study, the mean operative time is slightly higher than reported in the literature; however, no major intraoperative complications were observed.
Blood loss is difficult to assess. Some studies report intraoperative blood loss, while others emphasize the need for transfusions as an indirect parameter of blood loss. Some studies report transfusion rates over 50% after DFR (26,27); others described minimized bleeding (24). Similarly, in this study, bleeding was relatively limited, with 4 of 14 patients transfused (28.6%) and no patients requiring intensive transfusion treatments after surgery.
Literature-reported LOS varies widely from 6 to 26 days (24,26-28). In this study, the reported LOS was 19.9 days ranging from 9 to 63 days. LOS was thus largely variable among patients undergoing DFR at our institution. This finding could be explained by the patients’ complex background, comorbidity, and invasive procedure, which resulted in variable and often long-lasting LOS.
The main advantage of DFR is the knee’s early mobilization with free ROM and full weight-bearing from the first postoperative day, preserving the articular function and preventing the risk of prolonged bed rest (29). There is a wide range in the literature regarding PROMs after DFR: Angers-Goulet et al., in their case series, reported a mean postoperative KSS knee score of 42 and KSS function score of 60, while Vertesich et al. found a mean postoperative KSS knee score of 60 and KSS function score of 20 (30,31). Vaishya et al. and Matar et al. reported mean postoperative KSS knee scores of 78 and 80, respectively (21,22). The mean KSS knee score and OKS score described in our study are within this range and in line with those reported by Meluzio et al. in their systematic review (23).
The literature reports several intraoperative and postoperative complications ranging from 7% to 36% (22,25). The most frequent one is periprosthetic joint infection (PJI) which may require deep debridement, mobile parts replacement, prolonged antibiotic therapy, and sometimes limb amputation; the second one is aseptic component loosening (21,25,30). Periprosthetic fractures, extensor mechanism injuries and patellar maltracking, peroneal nerve palsy, bleeding and delayed wound healing are also widely described as DFR-related complications (24).
In our study, only one patient had a PJI that led to limb amputation after several salvage procedures, such as deep debridement and prolonged antibiotic therapy. The other complications were two delayed wound healing requiring surgical debridement and four cases of anemia. No deaths were reported at the last FU, while the literature describes a high incidence of mortality (14,32). This finding could be partially explained by the study’s relatively short FU period and the attention to the postoperative rehabilitation program.
Finally, it should be noted that ORIF and RIMN are often considered more conservative treatments than DFR. However, some studies comparing ORIF and RIMN show a slow postoperative recovery with no weight-bearing permission and restricted knee ROM (33,34). Moreover, other studies comparing DFR and ORIF show no differences in mortality and complication rates (14,32) and comparable postoperative PROMs (11), thus suggesting DFR could be a viable option in DFF and PDFF in elderly patients.
This study has several strengths. First, the study findings align with those reported in the literature, thus suggesting a certain reliability. Second, the results on PROMs and mortality provide some confidence concerning the feasibility of DFR after DFFs and PDFFs in elderly patients. Third, all the reported complications were successfully treated except one. This could be considered a good outcome in terms of complications management. Fourth, in our institution, patients diagnosed with PDFF were examined using three different classification systems, and each classification provides helpful information for decision-making.
This study also has several limitations. First, it is a retrospective study, therefore, has all the intrinsic limitations due to its design. Second, the small patients’ number could influence the strength of the results. Third, the patients lost at FU could represent a selection bias. Fourth, different megaprosthesis implants were employed on the patients selected in the study: this could also be a bias concerning the results. Further studies with more patients and a longer FU are needed to fully understand the feasibility and reliability of DFR implants after DFFs or PDFF in terms of postoperative PROMs and complications, including mortality.
Conclusions
Megaprosthesis implant in DFFs and PDFFs seems feasible, especially in elderly patients with previous symptomatic knee osteoarthritis, poor bone quality, multi-fragmentary fractures, or patients with loosening of the femoral prosthetic component. DFR could ensure a rapid return to walking, preserving knee function and reducing complications due to prolonged bed rest. However, a high complication rate and a lack of limb salvage procedures are reported; therefore, careful patient selection should be performed.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://aoj.amegroups.com/article/view/10.21037/aoj-23-21/rc
Data Sharing Statement: Available at https://aoj.amegroups.com/article/view/10.21037/aoj-23-21/dss
Peer Review File: Available at https://aoj.amegroups.com/article/view/10.21037/aoj-23-21/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aoj.amegroups.com/article/view/10.21037/aoj-23-21/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Institutional Review Board (IRB) of the CTO Hospital of Turin defined this study as exempt from IRB approval (retrospective study on a well-established surgical procedure) and conducted in accordance with the Declaration of Helsinki (as revised in 2013). All patients were informed about the study and consented to participate.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Canton G, Ratti C, Fattori R, et al. Periprosthetic knee fractures. A review of epidemiology, risk factors, diagnosis, management and outcome. Acta Biomed 2017;88:118-28. [PubMed]
- Benkovich V, Klassov Y, Mazilis B, et al. Periprosthetic fractures of the knee: a comprehensive review. Eur J Orthop Surg Traumatol 2020;30:387-99. [Crossref] [PubMed]
- Kuzyk PRT, Watts E, Backstein D. Revision Total Knee Arthroplasty for the Management of Periprosthetic Fractures. J Am Acad Orthop Surg 2017;25:624-33. [Crossref] [PubMed]
- Elsoe R, Ceccotti AA, Larsen P. Population-based epidemiology and incidence of distal femur fractures. Int Orthop 2018;42:191-6. [Crossref] [PubMed]
- Lesh ML, Schneider DJ, Deol G, et al. The consequences of anterior femoral notching in total knee arthroplasty. A biomechanical study. J Bone Joint Surg Am 2000;82:1096-101. [Crossref] [PubMed]
- Zalzal P, Backstein D, Gross AE, et al. Notching of the anterior femoral cortex during total knee arthroplasty characteristics that increase local stresses. J Arthroplasty 2006;21:737-43. [Crossref] [PubMed]
- Rice OM, Springer BD, Karunakar MA. Acute Distal Femoral Replacement for Fractures About the Knee in the Elderly. Orthop Clin North Am 2020;51:27-36. [Crossref] [PubMed]
- Quinzi DA, Childs S, Lipof JS, et al. The Treatment of Periprosthetic Distal Femoral Fractures After Total Knee Replacement: A Critical Analysis Review. JBJS Rev 2020;8:e2000003. [Crossref] [PubMed]
- Ebraheim NA, Kelley LH, Liu X, et al. Periprosthetic Distal Femur Fracture after Total Knee Arthroplasty: A Systematic Review. Orthop Surg 2015;7:297-305. [Crossref] [PubMed]
- Canton G, Tomic M, Giunta M, et al. Distal femur periprosthetic knee fractures in elderly patients: clinical and radiographic outcome after internal fixation. Acta Biomed 2021;92:e2021028. [PubMed]
- Hart GP, Kneisl JS, Springer BD, et al. Open Reduction vs Distal Femoral Replacement Arthroplasty for Comminuted Distal Femur Fractures in the Patients 70 Years and Older. J Arthroplasty 2017;32:202-6. [Crossref] [PubMed]
- Haidukewych GJ. Role of Distal Femoral Replacement for Periprosthetic Fractures Above a Total Knee Arthroplasty: When and How? J Orthop Trauma 2019;33:S33-5. [Crossref] [PubMed]
- Verma N, Jain A, Pal C, et al. Management of periprosthetic fracture following total knee arthroplasty- a retrospective study to decide when to fix or when to revise? J Clin Orthop Trauma 2020;11:S246-54. [Crossref] [PubMed]
- Darrith B, Bohl DD, Karadsheh MS, et al. Periprosthetic Fractures of the Distal Femur: Is Open Reduction and Internal Fixation or Distal Femoral Replacement Superior? J Arthroplasty 2020;35:1402-6. [Crossref] [PubMed]
- Lombardo DJ, Siljander MP, Sobh A, et al. Periprosthetic fractures about total knee arthroplasty. Musculoskelet Surg 2020;104:135-43. [Crossref] [PubMed]
- Rorabeck CH, Taylor JW. Periprosthetic fractures of the femur complicating total knee arthroplasty. Orthop Clin North Am 1999;30:265-77. [Crossref] [PubMed]
- Su ET, Kubiak EN, Dewal H, et al. A proposed classification of supracondylar femur fractures above total knee arthroplasties. J Arthroplasty 2006;21:405-8. [Crossref] [PubMed]
- Yoo JD, Kim NK. Periprosthetic Fractures Following Total Knee Arthroplasty. Knee Surg Relat Res 2015;27:1-9. [Crossref] [PubMed]
- Culliton SE, Bryant DM, MacDonald SJ, et al. Validity and Internal Consistency of the New Knee Society Knee Scoring System. Clin Orthop Relat Res 2018;476:77-84. [Crossref] [PubMed]
- Dawson J, Fitzpatrick R, Murray D, et al. Questionnaire on the perceptions of patients about total knee replacement. J Bone Joint Surg Br 1998;80:63-9. [Crossref] [PubMed]
- Vaishya R, Thapa SS, Vaish A. Non-neoplastic indications and outcomes of the proximal and distal femur megaprosthesis: a critical review. Knee Surg Relat Res 2020;32:18. [Crossref] [PubMed]
- Matar HE, Bloch BV, James PJ. Distal Femoral Replacements for Acute Comminuted Periprosthetic Knee Fractures: Satisfactory Clinical Outcomes at Medium-Term Follow-up. Arthroplast Today 2021;7:37-42. [Crossref] [PubMed]
- Meluzio MC, Oliva MS, Minutillo F, et al. The use of knee mega-prosthesis for the management of distal femoral fractures: A systematic review. Injury 2020;51:S17-22. [Crossref] [PubMed]
- Jassim SS, McNamara I, Hopgood P. Distal femoral replacement in periprosthetic fracture around total knee arthroplasty. Injury 2014;45:550-3. [Crossref] [PubMed]
- Sukhonthamarn K, Strony JT, Patel UJ, et al. Distal Femoral Replacement and Periprosthetic Joint Infection After Non-Oncological Reconstruction: A Retrospective Analysis. J Arthroplasty 2021;36:3959-65. [Crossref] [PubMed]
- Corap Y, Brix M, Emmeluth C, et al. Patient safety in distal femoral resection knee arthroplasty for non-tumor indications: a single-center consecutive cohort study of 45 patients. BMC Musculoskelet Disord 2022;23:199. [Crossref] [PubMed]
- Rahman WA, Vial TA, Backstein DJ. Distal Femoral Arthroplasty for Management of Periprosthetic Supracondylar Fractures of the Femur. J Arthroplasty 2016;31:676-9. [Crossref] [PubMed]
- Beckers G, Mazy D, Tollet P, et al. Knee mega-prosthesis in the management of complex knee fracture of the elderly: a case series and review of the literature. Acta Orthop Belg 2021;87:347-51. [Crossref] [PubMed]
- Khan S, Schmidt AH. Distal Femoral Replacement for Periprosthetic Fractures around Total Knee Arthroplasty: When and How? J Knee Surg 2019;32:388-91. [Crossref] [PubMed]
- Angers-Goulet M, Pelet S, Belzile EL, et al. Total knee arthroplasty with distal femoral replacement is associated with an important complication rate. A case series. Knee 2019;26:1080-7. [Crossref] [PubMed]
- Vertesich K, Puchner SE, Staats K, et al. Distal femoral reconstruction following failed total knee arthroplasty is accompanied with risk for complication and reduced joint function. BMC Musculoskelet Disord 2019;20:47. [Crossref] [PubMed]
- Hoellwarth JS, Fourman MS, Crossett L, et al. Equivalent mortality and complication rates following periprosthetic distal femur fractures managed with either lateral locked plating or a distal femoral replacement. Injury 2018;49:392-7. [Crossref] [PubMed]
- Shah JK, Szukics P, Gianakos AL, et al. Equivalent union rates between intramedullary nail and locked plate fixation for distal femur periprosthetic fractures - a systematic review. Injury 2020;51:1062-8. [Crossref] [PubMed]
- Horneff JG 3rd, Scolaro JA, Jafari SM, et al. Intramedullary nailing versus locked plate for treating supracondylar periprosthetic femur fractures. Orthopedics 2013;36:e561-6. [Crossref] [PubMed]
Cite this article as: Risitano S, D’Antonio D, Bosco F, Giustra F, Rocca F, Capella M, Sabatini L, Massè A. Should megaprosthesis implants be a viable option in elderly patients after distal femur and periprosthetic distal femur fractures?—a retrospective cohort study. Ann Joint 2023;8:33.


