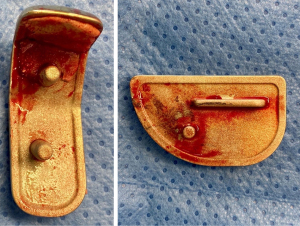
High early failure rate for a new unicondylar knee system
Introduction
Unicompartmental knee arthroplasty (UKA) is a procedure recognized worldwide for the treatment of unicompartmental femoro-tibial degeneration (1-3). It has been shown to be a satisfactory and less invasive alternative to TKA in selected patients (4,5). It is particularly attractive as an alternative to osteotomy or total knee arthroplasty (TKA), especially in middle-aged females (6).
Hypersensitivity to metallic implants remains relatively unpredictable and poorly understood, as well as a controversial topic among joint replacement surgeons (7,8). Coated implants have thus been developed to minimize the incidence of this complication (9).
In Germany over the last few decades an incidence of metal allergy of up to 20%—especially to nickel, cobalt and chromium—has been reported (10). This has prompted manufacturers to find ways to reduce allergic reactions in attempts to prolong implant survival. A new, hypoallergenic prosthesis, the Aesculap Univation (Aesculap®, Tuttlingen, Germany) with a corundum-blasting surface, has been used at the senior author’s institution since 2017. The basis for the design and surface coating of this prosthesis was to develop a low-friction system with anti-allergic coating to reduce the incidence of immune reactions, with theoretically prolonged survival. The corundum surface used in this new system is not a novel concept in itself. Previous authors published their results using ceramic surfaces and the prospects for their use in restorative bone and joint surgery since the 1980’s (11,12).
Here, we report our short-term experience with a new contemporary anti-allergic unicompartmental knee arthroplasty system which was specifically designed to address the issue of metal allergy. Given prior good results achieved using the anti-allergic Columbus Total Knee System for TKA, we started using this new UKA system believing that we would achieve similar favorable results. Our specific study objectives were (I) to estimate the rate of early failure with this new implant; and (II) to compare demographic features, morphometric, and clinical characteristics of patients experiencing versus not experience early prosthesis failure in terms of baseline demographic, morphometric, and clinical characteristics. We present the following article in accordance with the STROBE reporting checklist (available at https://aoj.amegroups.com/article/view/10.21037/aoj-22-18/rc).
Methods
Institutional board review approval
Our study obtained authorization from the Ethics Committee of our Hospital (Helios Klinik Berlin-Buch) for Human Research to carry out this study (No. 253/2021HKBB). A consent form was signed by each patient. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013).
Univation Aesculap UKA system
The Univation UKA is similar in its design to the Coated Columbus Knee System TKA (Aesculap®, Tuttlingen, Germany). To reduce ion release, the manufacturers added a multilayer coating system (Advanced Surface, AS) consisting of one thin adhesive chromium layer, five alternating intermediate layers composed of chromium nitride-chromium carbonitride (CrN-CrCN), and a final shielding layer of zirconium nitride (ZrN). This seven-layer coating system is applied to the surface of CoCrMo knee implants using a physical vapor deposition (PVD) method to achieve a total thickness of 4 µm (9). Therefore, there was no design change in the previous unicompartmental prosthesis. The manufacturer added the corundum surface to the previously developed unicompartment implant.
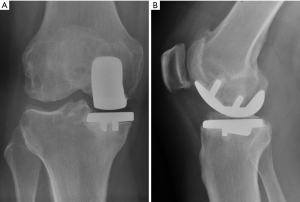
The current series was restricted to patients undergoing medial cemented UKA. Our indications for UKA were isolated unicompartmental OA or osteonecrosis; coronal deformity <15°; fixed flexion deformity <15°; intact anterior cruciate ligament and peripheral ligaments of the knee, as well as absence of inflammatory arthropathy. All patients had bone on bone changes in the medial compartment (13). In our study, there were no indications for UKA in the lateral compartment, as described by other surgeons (14,15). all procedures were performed using a similar technique. A fixed bearing cemented UKA prosthesis was used in all cases.
Between May 2017 and February 2020, 87 medial UKA procedures were performed in 82 patients. All patients had medial compartment disease caused by osteoarthritis, with no instances of osteonecrosis. In all cases, we used a cemented Univation hypoallergenic Aesculap® UKA. All procedures were performed at our institution by one of five experienced, fellowship-trained adult joint reconstruction surgeons (Figure 1).
Imaging protocol
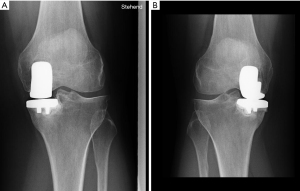
At our institution, standard radiographs are used routinely for patient diagnosis, surgical planning, and post-operative follow-up. These include antero-posterior, lateral, axial, panoramic and axial weight-bearing views. MediCAD® software is used to assist with planning every prosthesis implant procedure. All exams are recorded in our system. All cases were discussed at dedicated clinical meetings attended by at least three of the senior surgeons who performed the surgeries, as well as Radiology reports were reviewed by the Head of that Department. Thus, there was a consensus on the diagnoses (Figure 2).
Initial surgery
Preoperative templating was performed on all patients by the operating surgeon. During surgery, the patient was placed in a supine position and administered intravenous prophylactic antibiotics and tranexamic acid to reduce bleeding. A tourniquet was applied and inflated just before cementing. A medial parapatellar approach was performed in all cases.
Tibial cementation and insertion of the tibial component was performed prior to insertion of the femoral component in all cases. Third-generation cementing was consistently applied. Special care was taken to ensure a clean surgical field, including pulsatile jet lavage, followed by careful drying of all bone surfaces prior to cement application. Cement was applied under pressurized conditions. In all cases, pre-manufactured, antibiotic loaded PMMA bone cement (i.e., polymethylmethacrylate Palacos Gentamicin; Heraus Medical, Wehrheim, Germany) was used.
Given published results documenting safe weight-bearing and mobilization within one day in appropriately-selected patients (16), full weight bearing mobilization was permitted on the first post-operative day.
Revision surgery
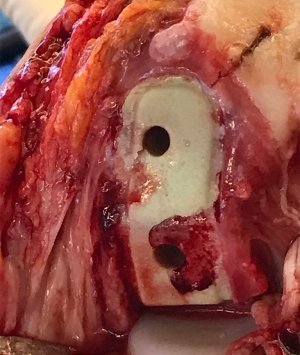
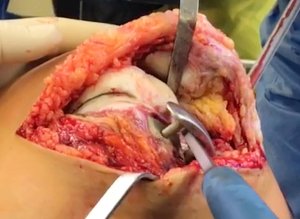
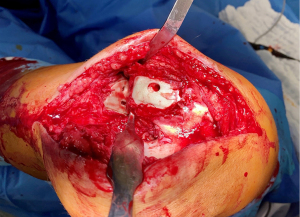
Prior to revision, all patients underwent knee joint aspiration to rule out infection. Following published guidelines derived from the 2018 International Consensus Meeting on Musculoskeletal Infections (17,18), all appropriate methods to diagnose prosthetic joint infection were undertaken, both to guide treatment and to identify infection as a possible cause of early prosthesis joint loosening. During revision surgery, microbiology samples again were collected, per standard surgical protocol (Figures 3-5).
Statistical analysis
In this paper, all continuous variables are reported as means with standard deviations (SD) ranges, while all categorical variables are reported as mean percentages, with 95% confidence intervals and ranges. Bivariate inter-group comparisons (between patients with versus without prosthesis failure) of continuous variables were conducted using Students t or non-parametric (log rank) tests, as indicated, while comparisons of categorical variables were conducted using Pearson χ2 analysis or Fisher’s exact test, as indicated. Survival analysis was performed to estimate the cumulative hazard of prosthesis failure through three years. All inferential tests were two-tailed with P≤0.05 set as the a-priori criterion for statistical significance. Statistical analysis was performed using statistical software R version 4.1.0. Packages used for survival analyses were survival version 3.2.11 and survminer version 0.4.9.
Results
Over the period of observation, UKA was performed on 87 joints in 82 patients. Demographic data is shown in Table 1.
Table 1
| Characteristic | 87 subjects, n (%) |
|---|---|
| Gender | |
| Male | 37 (43%) |
| Female | 50 (57%) |
| Age, years | 61 (11) |
| BMI, mean ± standard deviation | 29.8 (3.9) |
| Smoker | 20 (25%) |
| Unknown | 7 |
| Joint side | |
| Left | 47 (54%) |
| Right | 40 (46%) |
| Post-op outcome | |
| Success | 67 (77%) |
| Failure | 20 (23%) |
BMI, body mass index.
Twenty of the 87 implants (23%) failed, eight in males and 12 in females. The average age of the patients who experienced early prosthesis failure was 59.3 years (range 41–84 years) and the mean BMI was 29.8% (range 21.2–40.8). Two of these 20 patients were smokers. The earliest onset of symptoms was three months post-operatively. All 20 patients presented complaining of severe pain and swelling, and of progressively reduced range of motion in the affected knee.
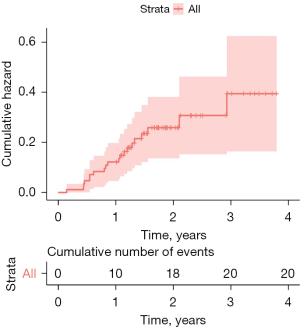
The mean time between UKA surgery and the diagnosis of prosthesis loosening diagnose was 13 months (5–29 months). The cumulative three-year failure rate was 33% (95% CI: 15–46%). After one year under observation, the rate of prosthesis failure was 12%, increasing to 23% within two years. The follow-up was three years (Table 2). Comparing patients who experienced prosthetic joint failure against those who did not, there were no significant differences in age, sex, height, weight, BMI, preoperative or postoperative AP angle, the amount of correction obtained, posterior tibial slope obtained, or the angle of the femoral component in the sagittal plane (Figures 6,7).
Table 2
| Characteristic | 3-year prosthetic loosening | P value |
|---|---|---|
| Overall | 33% (15–47%) | |
| Gender | >0.9 | |
| Male | 36% (3.3–58%) | |
| Female | 33% (8.7–50%) | |
| Smoker | 0.3 | |
| No | 25% (10–37%) | |
| Yes | 63% (0–91%) | |
| Joint | >0.9 | |
| Left | 28% (10–43%) | |
| Right | 39% (0.9–62%) |
For P values, groups were compared using the Log-rank test.
Discussion
Unicompartmental knee replacement surgery has produced encouraging results using various implant designs and techniques in recent decades. Implant survival rates of over 90% have been reported at up to 10 to 15 years after surgery (19-21). Given the increased interest of patients in Germany and generally increasing discussion regarding anti-allergic implants, we started using an antiallergic UKA system in 2017. The first ceramic implant for unicondylar tibial surface replacement was reported 50 years ago and resulted in limited wear between the ceramic and cartilaginous surfaces (22). Undercuts generated is a highly efficient way to augment mechanical adhesion to a Co-Cr-Mo surface; however, this is not possible on a ceramic surface, due to the brittleness of ceramics. Lack of bone cement retention promotes micromotion of the prostheses which, in turn, predictably leads to early aseptic loosening (23).
Following recognition of this high rate of prosthesis failure (projected as 33% over the first three years), we immediately stopped using the implant. We also sent an official warning to the manufacturer of this implant. Consequently, all further distribution of the anti-allergic UKA system was promptly discontinued in Germany to this day.
So far, the main cause of this extremely high rate of early aseptic loosening is not yet clear. A comparable total knee system using the same coating and produced by the same company has shown stable and satisfactory results (24). Some authors describe allergic reactions to the constituent metal used for the metallic components of the prostheses. Apostolopoulos et al. (25) described severe evidence of metallosis within the periprosthetic soft tissues of a 67-year-old female patient. In this patient, the tibial component was found to be loose, and the polyethylene bearing dislocated posteriorly. The same authors performed revision surgery for UKA failure due to allergic reactions to a TKA using an oxinium implant, identifying a positive reaction to a nickel lymphocyte proliferation skin allergy test (26). A similar case was reported by Bergschmidt et al. (27), involving a 58-year-old female patient with type IV hypersensitivity against both the nickel-II-sulfate and palladium chloride used during a TKA.
Law et al. (8) performed a retrospective review of a cohort of patients with self-reported metal allergy who underwent primary TKA employing an alternative ion-impregnated titanium implant. After a 4.6 years of follow-up, their results were encouraging, suggesting a potential implant option for patients with self-reported metal sensitivity. It is precisely this type of allergic reaction that we try to avoid with the newly-conceived hypoallergenic systems. Most of our 20 patients who experienced prosthetic joint failure were discovered to have synovial membrane thickening with hyperemia, an exudate, and synovium interposed between the cement and prosthesis surfaces at the time of revision. However, the patients’ skin showed no signs of allergy such as petechiae, dermatitis, or pruritis. For this reason, our impression is that the early loosening we observed was not an allergic reaction.
Similar to Mariani et al. (28), we were unable to identify any factors that were statistically associated with early loosening of the femoral component. There was no statistical association between early loosening of the femoral component and age, sex, height, weight, BMI, preoperative or postoperative AP angle, the amount of correction obtained, posterior tibial slope obtained, or the angle of the femoral component in the sagittal plane.
In our cohort, loosening of the contact surface between the metal components and cement mantle—which remained fixed in most cases—was the most prevalent factor (Figures 7,8). The quality of cementation was similar to what we have observed over decades during other TKA and THA our team has performed. As such, we do not feel that the high rate of loosening we observed was due to technical deficiencies, since we have never experienced such levels of failure with our other total joint surgeries.
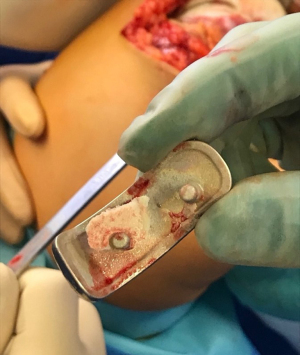
The modes of UKA failure most commonly reported over the first and second decades post implantation have been polyethylene wear, progressive arthritis, and component loosening (29), none of which we observed in our series. Campbell et al. (30) reported the failure of biconcave unicompartmental polyethylene developed for mobile-bearing UKA, including four polyethylene fractures in the central area. The system they used was thereafter removed from the market. Arastu et al. (31) also noted failure with mobile bearings, reporting a 21% rate of required revision for the LCS Preservation mobile-bearings prostheses within 22 months of implantation. The high failure rate identified in this study led to them ceasing to use this implant. Citak et al. (32) and Epinette et al. (33) reported similar results, with more than 50% of revisions failing in both series within the first five years after UKA implantation. We did not attempt to model past three years; but it certainly is conceivable that our patients would have experienced at least a 50% failure rate over two additional years of implant wear.
Other surgeons have experienced the failure of ceramic-coated implants manufactured by the same company (Aesculap) that manufactures the Universion UKA. Lionberger et al. (34) also observed an alarming rate of cement debonding with tibial implants. As with our cases, these authors reported how easily the implant could be lifted out of its cement bed, leaving a perfect implant imprint indicating the previous fixation point.
In the National Joint Registry (NJR) of England and Wales, reasons for UKA revision include infection (6% of cases), loosening/lysis (30%), component/polyethylene wear (2%), intractable pain (23%), and progressive osteoarthritis (4%), among others. In their last three-year period report, out of 33,676 primary UKA, unicondylar knee surgery involved using a mobile bearing in 62.5% (35).
In their twenty-year report, the New Zealand Joint Registry identified 1,038 revisions among 12,627 registered UKA (8%). The mean time between UKA and revision was 2,080 days, with a minimum of 4 days. In the first year, 13.9% of revisions were caused by pain, 9.3% by femoral component loosening, and 17.7% by tibial component loosening (36).
Reporting on the results of a 25-center study, Epinette et al. noted that 19% of the UKA revisions occurred within the first year and 48.5% within the first five years. Loosening was the main reason for failure (45%), followed by osteoarthritis progression (15%) and wear (12%) (33).
The results that most closely resemble our own were reported by Mariani et al. (28), who diagnosed early failure in 39 UKAs, 15 (38%) from 9 to 12 months postoperatively. All patients in their series had received a DePuy Preservation prosthesis (DePuy, Warsaw, In) with an all-poly tibial component.
Our study has several limitations, foremost among them the lack of a control group. Second, at the beginning of the study, we were not expecting such a substantial percentage of early failure with this new device. Thus, we faced the dilemma of deciding which patient and procedural parameters were most important after our series had begun. Third, our evaluation was limited to macroscopic reasons for UKA loosening, without exploring potential microscopic explanations. For the latter, we addressed our concerns to the manufacturer of this prosthesis, assuming that they will be compelled to perform their own evaluations before reintroducing this or any similar prosthesis into the market.
Conclusions
In our series of 87 procedures using an anti-allergic UKA system, we observed an unexpectedly and unacceptably high rate of early loosening, projected to reach roughly one in three cases within three years. Though no cause for this high rate of prosthesis failure was identified, it is our belief that the answer lies in the loss of bonding at the implant-cement-bone interface. Until the reason is found, and the device modified to correct for this shortcoming, we have stopped using and the manufacturer has ceased distributing this implant.
Acknowledgments
We would like to thank Prof. Felipe Figueiredo, consultant in Biostatistics and Epidemiology, who is essential in data analysis and statistical calculations for this study.
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://aoj.amegroups.com/article/view/10.21037/aoj-22-18/rc
Data Sharing Statement: Available at https://aoj.amegroups.com/article/view/10.21037/aoj-22-18/dss
Peer Review File: Available at https://aoj.amegroups.com/article/view/10.21037/aoj-22-18/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://aoj.amegroups.com/article/view/10.21037/aoj-22-18/coif). DK serves as an unpaid editorial board member of Annals of Joint from April 2022 to March 2024. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Our study obtained authorization from the Ethics Committee of our Hospital (Helios Klinik Berlin-Buch) for Human Research to carry out this study (No. 253/2021HKBB). A consent form was signed by each patient. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Vasso M, Antoniadis A, Helmy N. Update on unicompartmental knee arthroplasty: Current indications and failure modes. EFORT Open Rev 2018;3:442-8. [Crossref] [PubMed]
- Kagan R, Anderson MB, Bailey T, et al. Ten-Year Survivorship, Patient-Reported Outcomes, and Satisfaction of a Fixed-Bearing Unicompartmental Knee Arthroplasty. Arthroplast Today 2020;6:267-73. [Crossref] [PubMed]
- Riebel GD, Werner FW, Ayers DC, et al. Early failure of the femoral component in unicompartmental knee arthroplasty. J Arthroplasty 1995;10:615-21. [Crossref] [PubMed]
- Saldanha KA, Keys GW, Svard UC, et al. Revision of Oxford medial unicompartmental knee arthroplasty to total knee arthroplasty - results of a multicentre study. Knee 2007;14:275-9. [Crossref] [PubMed]
- Bruce DJ, Hassaballa M, Robinson JR, et al. Minimum 10-year outcomes of a fixed bearing all-polyethylene unicompartmental knee arthroplasty used to treat medial osteoarthritis. Knee 2020;27:1018-27. [Crossref] [PubMed]
- Bouguennec N, Mergenthaler G, Gicquel T, et al. Medium-term survival and clinical and radiological results in high tibial osteotomy: Factors for failure and comparison with unicompartmental arthroplasty. Orthop Traumatol Surg Res 2020;106:S223-30. [Crossref] [PubMed]
- Thienpont E, Berger Y. No allergic reaction after TKA in a chrome-cobalt-nickel-sensitive patient: case report and review of the literature. Knee Surg Sports Traumatol Arthrosc 2013;21:636-40. [Crossref] [PubMed]
- Law JI, Morris MJ, Hurst JM, et al. Early Outcomes of an Alternative Bearing Surface in Primary Total Knee Arthroplasty in Patients with Self-reported Metal Allergy. Arthroplast Today 2020;6:639-43. [Crossref] [PubMed]
- Lützner J, Hartmann A, Dinnebier G, et al. Metal hypersensitivity and metal ion levels in patients with coated or uncoated total knee arthroplasty: a randomised controlled study. Int Orthop 2013;37:1925-31. [Crossref] [PubMed]
- Thomas P, Schuh A, Ring J, et al. Joint statement by the Implant Allergy Working Group (AK 20) of the DGOOC (German Association of Orthopedics and Orthopedic Surgery), DKG (German Contact Dermatitis Research Group) and DGAKI (German Society for Allergology and Clinical Immunology). Hautarzt 2008;59:220-9. [Crossref] [PubMed]
- Korzh AA, Degtiareva EV, Gruntovskiĭ GKh. Corundum ceramics and the prospects for their use in restorative bone and joint surgery. Ortop Travmatol Protez 1981;5-8.
- Gudushauri ON, Dumbadze GG, Mikadze GS. Results and prospects of using endoprostheses made of corundum ceramics. Khirurgiia (Mosk) 1985;124-8.
- KELLGREN JH. LAWRENCE JS. Radiological assessment of osteo-arthrosis. Ann Rheum Dis 1957;16:494-502. [Crossref] [PubMed]
- Volpi P, Marinoni L, Bait C, et al. Lateral unicompartimental knee arthroplasty: indications, technique and short-medium term results. Knee Surg Sports Traumatol Arthrosc 2007;15:1028-34. [Crossref] [PubMed]
- Citak M, Cross MB, Gehrke T, et al. Modes of failure and revision of failed lateral unicompartmental knee arthroplasties. Knee 2015;22:338-40. [Crossref] [PubMed]
- Gondusky JS, Choi L, Khalaf N, et al. Day of surgery discharge after unicompartmental knee arthroplasty: an effective perioperative pathway. J Arthroplasty 2014;29:516-9. [Crossref] [PubMed]
- Schwarz EM, Parvizi J, Gehrke T, et al. 2018 International Consensus Meeting on Musculoskeletal Infection: Research Priorities from the General Assembly Questions. J Orthop Res 2019;37:997-1006.
- Shohat N, Bauer T, Buttaro M, et al. Hip and Knee Section, What is the Definition of a Periprosthetic Joint Infection (PJI) of the Knee and the Hip? Can the Same Criteria be Used for Both Joints?: Proceedings of International Consensus on Orthopedic Infections. J Arthroplasty 2019;34:S325-7. [Crossref] [PubMed]
- Camanho GL. UNICOMPARTIMENTAL KNEE ARTHROPLASTY - 15 YEARS FOLLOW UP. Acta Ortop Bras 2020;28:233-5. [Crossref] [PubMed]
- Neufeld ME, Albers A, Greidanus NV, et al. A Comparison of Mobile and Fixed-Bearing Unicompartmental Knee Arthroplasty at a Minimum 10-Year Follow-up. J Arthroplasty 2018;33:1713-8. [Crossref] [PubMed]
- Berger RA, Meneghini RM, Jacobs JJ, et al. Results of unicompartmental knee arthroplasty at a minimum of ten years of follow-up. J Bone Joint Surg Am 2005;87:999-1006. [Crossref] [PubMed]
- Langer G. Ceramic Tibial Plateau of the 70s. Bioceramics in Joint Arthroplasty. 2002;7th International Biolox Symposium.
- Marx B, Kerschbaum P, Lindlahr S, et al. Bone Cement Adhesion on Ceramic Surfaces - Deactivation of Surfaces and as a Consequence Inefficient Retention of Knee Prostheses Because of Adsorption of Atmospheric Water. Z Orthop Unfall 2018;156:85-92. [Crossref] [PubMed]
- Goebel D, Schultz W. The Columbus Knee System: 4-Year Results of a New Deep Flexion Design Compared to the NexGen Full Flex Implant. Arthritis 2012;2012:213817. [Crossref] [PubMed]
- Apostolopoulos AP, Katsougrakis I, Fanous R, et al. Severe metallosis following polyethylene dislocation in a mobile-bearing medial unicompartmental knee replacement. J Long Term Eff Med Implants 2014;24:147-50. [Crossref] [PubMed]
- Apostolopoulos AP, Balfousias T, Khan S, et al. Failure of a Medial Unicompartmental Knee Replacement due to Metal Allergy. J Long Term Eff Med Implants 2018;28:319-25. [Crossref] [PubMed]
- Bergschmidt P, Bader R, Mittelmeier W. Metal hypersensitivity in total knee arthroplasty: revision surgery using a ceramic femoral component - a case report. Knee 2012;19:144-7. [Crossref] [PubMed]
- Mariani EM, Bourne MH, Jackson RT, et al. Early failure of unicompartmental knee arthroplasty. J Arthroplasty 2007;22:81-4. [Crossref] [PubMed]
- Aleto TJ, Berend ME, Ritter MA, et al. Early failure of unicompartmental knee arthroplasty leading to revision. J Arthroplasty 2008;23:159-63. [Crossref] [PubMed]
- Campbell D, Lewis P, Mooney L. Catastrophic failure of biconcave unicompartmental polyethylene bearings. Knee 2020;27:987-92. [Crossref] [PubMed]
- Arastu MH, Vijayaraghavan J, Chissell H, et al. Early failure of a mobile-bearing unicompartmental knee replacement. Knee Surg Sports Traumatol Arthrosc 2009;17:1178-83. [Crossref] [PubMed]
- Citak M, Dersch K, Kamath AF, et al. Common causes of failed unicompartmental knee arthroplasty: a single-centre analysis of four hundred and seventy one cases. Int Orthop 2014;38:961-5. [Crossref] [PubMed]
- Epinette JA, Brunschweiler B, Mertl P, et al. Unicompartmental knee arthroplasty modes of failure: wear is not the main reason for failure: a multicentre study of 418 failed knees. Orthop Traumatol Surg Res 2012;98:S124-30. [Crossref] [PubMed]
- Lionberger D, Conlon C, Wattenbarger L, et al. Unacceptable failure rate of a ceramic-coated posterior cruciate-substituting total knee arthroplasty. Arthroplast Today 2019;5:187-92. [Crossref] [PubMed]
- Baker PN, Petheram T, Avery PJ, et al. Revision for unexplained pain following unicompartmental and total knee replacement. J Bone Joint Surg Am 2012;94:e126. [Crossref] [PubMed]
- Association NZO. The New Zealand Joint Registry. Twenty year report: January 1999-December 2018. Available online: https://www.nzoa.org.nz/sites/default/files/NZJR_22_Year_Report_Final.pdf
Cite this article as: Toledo de Araujo LC, Freitag J, Kamath AF, Sandiford NA, Kendoff D. High early failure rate for a new unicondylar knee system. Ann Joint 2023;8:3.