
Patellofemoral knee arthroplasty
Introduction
Isolated patellofemoral Joint Osteoarthritis (PFOA) is a common degenerative disease involving 10–25% of patients with symptomatic knee osteoarthritis (OA) (1-4), Noble et al. reported an incidence of 79% in a cadaveric study on patients aged over 65 years (5). PFOA is by far more common than isolated medial or lateral femorotibial osteoarthritis (FTOA). An association with symptomatic medial FTOA was identified in 12% of patients (3). The lateral facet is more frequently involved than the medial one (92% vs. 8%) (6).
McAlindon et al. observed an augmented prevalence in females for isolated anterior compartment degeneration (24% vs. 11%) (2) but a recent systematic review and meta-analysis revealed comparable prevalence in both sexes (27% vs. 26%) (4). This lack of consensus can be expression of the variable entity of symptoms and by the fact that the progression of the degenerative process is typically slow (7). In presence of symptomatic high grade PFOA prosthetic treatment is generally considered as the gold standard of treatment. Despite this general consideration there is still a lack of consensus about the role of patellofemoral arthroplasty (PFA). The results reported in literature based on first generation PFA lead some authors to consider this procedure as a bridge before total knee replacement or even to contraindicate PFA in favor of total knee arthroplasty (TKA) (8,9). Nevertheless, improvement in new generation prosthetic design and surgical techniques demonstrated more consistent clinical results and good mid-term implant survivorship bringing more surgeons to consider this procedure as safe and effective in treating isolated PFOA (10).
Aetiology and classification
PFOA can recognize different aetiologies such as previous recurrent patellar dislocation, cartilage lesions, inflammatory diseases and patellar fractures sequelae. Dejour and Allain in a multicentric study identified four groups possibly leading to PFOA: 8% were concomitant to inflammatory diseases such as chondrocalcinosis, 9% were associated to previous patellar fracture, a large group (33%) was associated to previous patellofemoral dislocations and finally 49% were considered as primary (idiopathic) (7).
The most common classification system is the one described by Iwano et al. (6), based on axial view X-rays. Four stages are described:
- Stage 1: patellofemoral remodelling;
- Stage 2: reduction on joint line less than 3 mm;
- Stage 3: reduction of joint line more than 3 mm;
- Stage 4: bone on bone facet.
Predisposing factors
Anatomical abnormalities are described as a potential predisposing factor in developing of PFOA. The most common abnormalities are: patella alta, excessive patellar tilt, off-set and trochlear dysplasia (11-13). These considerations were confirmed by Dejour and Allain who observed radiological signs of trochlear dysplasia in 78% of isolated PFOA (7). One of the possible explanations is that supratrochlear spur, identifiable in type B and D trochlear dysplasia (14), could increase patellofemoral joint load in flexion performing a so called “Anti-Maquet effect”. A correlation between patella dysplasia and PFOA was also observed, with PFOA occurring more frequently in Wiberg type 2 patellas (7,15).
Relationships between patellofemoral disorders in young population can be correlated with an increased risk of knee OA changes (16,17). Recurrent patellofemoral dislocation was recognized as one on the main causes by Conchie et al. reporting an increased risk (adjust OR of 3.2) of developing PFOA in presence of previous patellar dislocations. These data were similar to the one observed in patients who underwent previous knee surgeries. (adjust OR of 3.5) (16).
In a recent cohort study, including 609 patients, Sanders et al. observed an even higher risk of PFOA in subjects who experienced a lateral patellar dislocation (HR 7.8; 95% CI, 3.9–17.6; P<0.001) (17).
Treatment
Being most of isolated PFOA symptoms well tolerated, and knowing that clinical and radiological progression can be very low, the treatment of PFOA is mainly conservative (7).
In patients not responding to conservative treatment (rehabilitation program, weight reduction, oral pain killers or intrarticular viscosupplementation) (18-21), surgical treatment is a viable option in order to provide pain relief and restore PF joint function.
In treatment of PFOA many surgical procedures have been described, reporting variable results, varying from Lateral release (22), Anterior Tibial Tuberosity (ATT) osteotomy (23,24), reparative surgery of cartilage lesions (25), patellectomy (26), PFA (27-30) and TKA (8,9,31,32).
In advanced stage PFOA most of the authors advocate prosthetic solutions which include PFA and TKA. In the recent past PFA is gaining more and more popularity considering the promising results of new generation implants and less invasive surgical technique when compared to TKA.
Patellofemoral joint arthroplasty
McKeever first reported in 1955 the use of a screw-on Vitallium patellar implant in alternative to patellectomy, historically associated to unsuccessful clinical results (33). Despite having short-term good results, also confirmed by Levitt (34) and Vermeulen et al. (35), early failure of McKeever prosthesis was reported due to the rapid native trochlear wear (36). In 1979 a new generation implant, the Richards PFA, was presented but results were inconsistent (37). In the past few years the concept of PFA has been constantly evolving with introduction of new generation implants and surgical techniques.
Resurfacing implants
This kind of implants is meant to replace worn cartilage laying on subchondral bone. Being based on pre-existing anatomy of the native trochlea, anatomical parameters such as shape or orientation remain unchanged when using this kind of implants, the resurfacing implants are also defined as “Inlay”. This type of design allows a highly conservative bone preparation, flush with native surrounding cartilage and parallel to the original trochlear inclination. The mediolateral coverage of the trochlea is limited and the proximal extension of the component is contained by the trochlear cartilage surface.
Implant positioning highly depends on underlying trochlear anatomy, therefore the presence of trochlear dysplasia can lead to a greater risk of components malpositioning (in particular excessive internal-rotation) possible leading to instability or maltracking. In addiction surgical technique mainly relies on free hand preparation, leading to reduced reproducibility and accuracy in implants positioning. Associated surgical procedures are necessary in presence of excessive patella height and TT-TG (Tibial Tuberosity Tibial Groove) to achieve a proper patellar tracking.
Trochlear cutting implants
In 1990s the poor clinical results of first generation implants, mainly due to patellofemoral maltracking and instability, lead to the development of the second generation patellofemoral prosthesis, the so-called “Onlay design”These implants are based on TKA anterior flange, flush with the anterior femoral cortex, reducing the risk of potential impingement with residual trochlear bone (38,39).
Second generations implants are characterized by a wider trochlea in mediolateral size, extending more proximally than the original cartilage, a less obtuse sagittal radius of curvature, a higher level of patellar constraint and a high asymmetrical lateral flange, in order to resist lateral dislocation quadriceps force (40).
The surgical technique is more reproducible and adaptable to the patient’s anatomic characteristics, allowing a better rotational control of the femoral component whatever the pre-existing anatomy, therefore additional extensor mechanism procedures are no longer systematically required in order to obtain implant stability.
These advancements in components design lead to reduction of snapping, catching and popping, allowing free transverse movement in full extension.
Indications
The success of the surgical procedure heavily depends upon patient selection. Most of the authors agree on performing PFA in isolated PFOA, whatever the aetiology, greater than Iwano stage 2. Another typical indication is failure of previous surgeries such as ATT osteotomy or cartilage regeneration procedures. A BMI higher than 30 represents, according to van Jonbergen et al. (41), a relative contraindication. These findings are supported by Dahm et al. who reported significantly lower (P>0.03) KSS improvement in this category of patients despite not finding any significative difference in pain, knee flexion, satisfaction and progression of tibiofemoral joint OA (27).
Observing that progression of Osteoarthritis to the non-replaced tibiofemoral compartments is the main cause of revision in PFOA, presence of OA great than Kellgren-Lawrence grade 1 in medial or lateral compartment, excessive valgus or varus and history of meniscal surgery represent an important contraindication. Other absolute contraindications are Complex Regional Pain Syndrome, Inflammatory diseases such as, chondrocalcinosis and rheumatoid arthritis, active infections and psychogenic anterior knee pain (42-44).
Outcomes
PFA is considered to have several theoretical advantages if compared to TKA, such as good bone-stock and soft-tissue preservation (31), reduced blood loss and hospitalization time (33) and more physiological knee kinematics potentially allowing faster recovery and better clinical outcomes (Table 1).
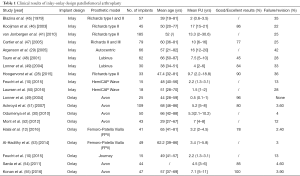
Full table
Despite these considerations about re-creating kinematics close to physiology, the number of biomechanical studies investigating this aspect is limited and results are heterogeneous (40,45,56).
Resurfacing implants
A large number of studies are available for first generation Inlay-design although heterogeneous in implanted prosthetic model, inclusion criteria and quality. Hoogervorst et al. reported good to excellent results in satisfaction score in 90% of patients, a survival rate of 73% at 10 years, with a TKA conversion rate of 21% at 5.5 years, mainly due to TFOA progression (71% of cases) (28). These data appear to be comparable to the ones reported by Van Jonbergen et al. (84% survival-rate at 10 years) (41) and Cartier et al. (75% survival rate at 10 years) (47).
Considering the 90% survival rate at 10 years, set as acceptable minimal threshold by ODEP benchmark for prosthetic implants, was not reached.
The author stressed the importance of carefully excluding TFOA, defined as the main cause of failure, in order to reduce the re-intervention rate after PFA (28).
Good to excellent long term results at a mean FU of 17.8±0.8 years were reported by Kooijman et al. showing a 98% survival rate and a satisfaction rate of 86% (46). These positive data are contrasting with the one observed by Argenson et al. reporting a cumulative survival rate of only 58% at 16 years FU (29).
Regarding new generation Inlay prosthesis Feucht et al. reported in a short-term matched-pair comparison study (mean FU 24 months) with Onlay implants, similar result in clinical scores (P>0.05) and implant survivorship but a slower TFOA progression in the Inlay group (10).
Comparable results about the same implant have been recently reported by Cotic et al., in the authors’ view the more physiological placement of the Inlay prosthesis could reduce the risk of patellar overstuffing leading to a lower progression of TFOA (57).
According to these authors these so called “second generation implants” could increase patellar stability, without altering soft tissue tension and reducing the risk of patellar overstuffing (58,59). A recent mid-term study by Laursen et al., based on similar prosthetic design, reported a high revision rate (28%) at 6 years FU, leading the author to consider this kind of implants as a temporary solution, with good to excellent results in short-term period for younger patients ineligible for TKA (50). There is a need of long-term clinical studies in order to better define clinical results and revision rate of new Inlay prosthesis design which could diverge from the first generation one, even in consideration of the wider inclusion criteria used in the older studies including patients with initial signs of TFOA.
Trochlear cutting implants
The outcomes of Onlay design Implants were discussed by several clinical studies.
Halai et al. reported results about 31 patients (mean age 65 years) affected by idiopathic PFOA with a mean follow-up of 3.2 years, describing 78% of patient satisfaction with a 97.6% survival rate (12).
Al-Hadithy et al. reported about 41 patients affected by isolated PFOA, evaluated at mean follow-up of 37 months, with improvement of Oxford Knee Score (from 19.7 to 37.7) and a 97% survival rate (53).
These results seem comparable to those found in prior studies in which the percentage of good to excellent results was superior to 90% (30,60).
A slightly lower survivorship rate (82%) was reported by Mont et al. on 37 patients (mean age 49 years) and mean follow-up of 7 years (52). Outcomes of PFA in post-traumatic PFOA were described by Konan et al. reporting results on 51 patients (mean age 57 years). At the mean FU of 7.1 years the median Oxford Knee Score was 38 with excellent reported pain relief and functional outcomes. Survivorship was 96.1% and only two revisions were reported (one for residual pain and one for TFOA progression) (57).
In patients presenting PFOA variably associated to medial and/or lateral compartment OA and with intact ligaments, some authors suggest the combinations of PFA with unicompartmental knee arthroplasty (UKA). Data in literature reporting clinical results of these procedures combined are few, even though outcomes seem to be promising (61-64).
Further long-term high quality studies are needed to assess the clinical benefits of this implant association, even in consideration of the good results of TKA in treatment of PFOA (Table 2).

Full table
Complications
A high failure risk and complication rate in PFA was previously reported although nowadays improvement in prosthetic design and careful patient selection lead to better clinical results, with a significant decrease in complications rate.
A recent systematic review by van der List et al., combining cohort studies and registries, analysed the main causes of failure in PFA.
Early failure was most commonly due to unexplained pain (31%), OA progression (24%) and patellar maltracking (14%) (69). The reason of unexplained pain can be found in both progression of OA or a specific prosthetic design issues (70-72).
The most common reasons of late failure are represented by OA progression (46%), aseptic loosening (18%) and pain (9%) (69).
An interesting finding is the association between OA progression and the indication to PFA: Argenson et al. reported a higher incidence in patients with primary PFOA (44%) than post-traumatic OA (17%) and patellar instability (14%) (29). Similar conclusions were proposed by Nicol et al. (73) and Dahm et al. (27). Other conditions, such as Infections and implant loosening, play nowadays a minor role in implants failure (69).
A meta-analysis by Monk et al. (74) reported decreasing in PFA complications rate in recent implants design (from 39% to 14%) although still remaining higher than TKA (7%); the authors focused on the geometrical design of the prosthesis therefore attributing the most of the failure mechanism to a mechanical problem (instability, loosening, malalignment). A careful analysis of the risk-benefit rate should be accomplished by the surgeon, in accordance with the patient, in order to maximize the clinical results reducing the risk of further surgical revision.
Conclusions
PFJA has been considered an alternative to TKA in patients with advanced symptomatic isolated OA.
In the last decades new prosthetic design, materials and surgical techniques were developed changing radically the vision about patellofemoral replacement. Literature reports heterogeneous evidences of clinical results, from the excellent long-term follow-up outcomes described by Kooijman (mean FU 17.8±0.8 years, 98% survival rate and 86% satisfaction rate) (46) to the poorer ones reported by Argenson et al. (mean FU 16 years, cumulative survival 58%) (29). Despite the high complication and revision rate observed in the first generation implants, the last clinical available studies show a trend towards improvement, reporting a PFA complication rate decreasing from 39% to 14% even though still remaining higher than TKA (7%) (74).
Nowadays there is still no consensus on the ideal trochlear design, even in consideration of the new generation Inlay models which showed similar result in clinical scores (P>0.05) and implant survivorship over Onlay models (10,52). Careful patients selection as been highlighted as the key of success but there is still no agreement over precise guidelines for indicating PFA Further high level long term and randomized controlled studies are needed to confirm good results of PFA. Surgeons have to weigh the long-term success rate of TKA with the good clinical results in short-midterm FU of PFA, which also do not interfere with further conversion to TKA (75).
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editor (Luigi Sabatini) for the series “Osteotomies and partial replacement in early osteoarthritis of the knee” published in Annals of Joint. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/aoj.2017.07.09). The series “Osteotomies and partial replacement in early osteoarthritis of the knee” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Davies AP, Vince AS, Shepstone L, et al. The radiologic prevalence of patellofemoral osteoarthritis. Clin Orthop Relat Res 2002;206-12. [Crossref] [PubMed]
- McAlindon TE, Snow S, Cooper C, et al. Radiographic patterns of osteoarthritis of the knee joint in the community: the importance of the patellofemoral joint. Ann Rheum Dis 1992;51:844-9. [Crossref] [PubMed]
- Duncan RC, Hay EM, Saklatvala J, et al. Prevalence of radiographic osteoarthritis--it all depends on your point of view. Rheumatology (Oxford) 2006;45:757-60. [Crossref] [PubMed]
- Kobayashi S, Pappas E, Fransen M, et al. The prevalence of patellofemoral osteoarthritis: a systematic review and meta-analysis. Osteoarthritis Cartilage 2016;24:1697-707. [Crossref] [PubMed]
- Noble J, Hamblen DL. The pathology of the degenerate meniscus lesion. J Bone Joint Surg Br 1975;57:180-6. [PubMed]
- Iwano T, Kurosawa H, Tokuyama H, et al. Roentgenographic and clinical findings of patellofemoral osteoarthrosis. With special reference to its relationship to femorotibial osteoarthrosis and etiologic factors. Clin Orthop Relat Res 1990;190-7. [PubMed]
- Dejour D, Allain J. Histoire naturelle de l’arthrose fémoro-patellaire isolée. Rev Chir Orthop Reparatrice Appar Mot 2004;90:89-93. [Crossref]
- Mont MA, Haas S, Mullick T, et al. Total knee arthroplasty for patellofemoral arthritis. J Bone Joint Surg Am 2002;84-A:1977-81. [Crossref] [PubMed]
- Thompson NW, Ruiz AL, Breslin E, et al. Total knee arthroplasty without patellar resurfacing in isolated patellofemoral osteoarthritis. J Arthroplasty 2001;16:607-12. [Crossref] [PubMed]
- Feucht MJ, Cotic M, Beitzel K, et al. A matched-pair comparison of inlay and onlay trochlear designs for patellofemoral arthroplasty: no differences in clinical outcome but less progression of osteoarthritis with inlay designs. Knee Surg Sports Traumatol Arthrosc 2015; [Epub ahead of print]. [Crossref] [PubMed]
- Stefanik JJ, Zumwalt AC, Segal NA, et al. Association between measures of patella height, morphologic features of the trochlea, and patellofemoral joint alignment: the MOST study. Clin Orthop Relat Res 2013;471:2641-8. [Crossref] [PubMed]
- Halai M, Ker A, Anthony I, et al. Femoro Patella Vialla patellofemoral arthroplasty: An independent assessment of outcomes at minimum 2-year follow-up. World J Orthop 2016;7:487-93. [PubMed]
- Kalichman L, Zhang Y, Niu J, et al. The association between patellar alignment and patellofemoral joint osteoarthritis features--an MRI study. Rheumatology (Oxford) 2007;46:1303-8. [Crossref] [PubMed]
- Dejour D, Reynaud P, Le Coultre B. Douleurs et Instabilité rotulienne. Essai de Classification. Med Hyg 1998;56:1466-71.
- Wiberg G. Roentgenographic and anatomic studies on the femoro-patellar joint. Acta Orthop Scand 1941;12:319-410. [Crossref]
- Conchie H, Clark D, Metcalfe A, et al. Adolescent knee pain and patellar dislocations are associated with patellofemoral osteoarthritis in adulthood: A case control study. Knee 2016;23:708-11. [Crossref] [PubMed]
- Sanders TL, Pareek A, Johnson NR, et al. Patellofemoral Arthritis After Lateral Patellar Dislocation: A Matched Population-Based Analysis. Am J Sports Med 2017;45:1012-7. [Crossref] [PubMed]
- Abdul-Hadi O, Parvizi J, Austin MS, et al. Nonsteroidal anti-inflammatory drugs in orthopaedics. J Bone Joint Surg Am 2009;91:2020-7. [PubMed]
- Clarke S, Lock V, Duddy J, et al. Intra-articular hylan G-F 20 (Synvisc) in the management of patellofemoral osteoarthritis of the knee (POAK). Knee 2005;12:57-62. [Crossref] [PubMed]
- Amin S, Baker K, Niu J, et al. Quadriceps strength and the risk of cartilage loss and symptom progression in knee osteoarthritis. Arthritis Rheum 2009;60:189-98. [Crossref] [PubMed]
- Conrozier T, Mathieu P, Schott AM, et al. Factors predicting long-term efficacy of Hylan GF-20 viscosupplementation in knee osteoarthritis. Joint Bone Spine 2003;70:128-33. [Crossref] [PubMed]
- Aderinto J, Cobb AG. Lateral release for patellofemoral arthritis. Arthroscopy 2002;18:399-403. [Crossref] [PubMed]
- Fulkerson JP, Becker GJ, Meaney JA, et al. Anteromedial tibial tubercle transfer without bone graft. Am J Sports Med 1990;18:490-6. [Crossref] [PubMed]
- Kanazawa H, Maruyama Y, Shitoto K, et al. Survival and clinical results of a modified "crosse de hockey" procedure for chronic isolated patellofemoral joint osteoarthritis: mid-term follow-up. J Orthop Traumatol 2017;18:23-30. [Crossref] [PubMed]
- Schüttler S, Andjelkov N. Periosteal Transplantation Combined with the Autologous Matrix-Induced Chondrogenesis (AMIC) Technique in Isolated Patellofemoral Osteoarthritis: A Case Report. Cartilage 2012;3:194-8. [Crossref] [PubMed]
- Asopa V, Willis-Owen C, Keene G. Patellectomy for osteoarthritis: a new tension preserving surgical technique to reconstruct the extensor mechanism with retrospective review of long-term follow-up. J Orthop Surg Res 2015;10:107. [Crossref] [PubMed]
- Dahm DL, Kalisvaart MM, Stuart MJ, et al. Patellofemoral arthroplasty: outcomes and factors associated with early progression of tibiofemoral arthritis. Knee Surg Sports Traumatol Arthrosc 2014;22:2554-9. [Crossref] [PubMed]
- Hoogervorst P, de Jong RJ, Hannink G, et al. A 21% conversion rate to total knee arthroplasty of a first-generation patellofemoral prosthesis at a mean follow-up of 9.7 years. Int Orthop 2015;39:1857-64. [Crossref] [PubMed]
- Argenson JN, Flecher X, Parratte S, et al. Patellofemoral arthroplasty: an update. Clin Orthop Relat Res 2005;50-3. [Crossref] [PubMed]
- Odumenya M, Costa ML, Parsons N, et al. The Avon patellofemoral joint replacement: Five-year results from an independent centre. J Bone Joint Surg Br 2010;92:56-60. [Crossref] [PubMed]
- Odumenya M, McGuinness K, Achten J, et al. The Warwick patellofemoral arthroplasty trial: a randomised clinical trial of total knee arthroplasty versus patellofemoral arthroplasty in patients with severe arthritis of the patellofemoral joint. BMC Musculoskelet Disord 2011;12:265. [Crossref] [PubMed]
- Dahm DL, Al-Rayashi W, Dajani K, et al. Patellofemoral arthroplasty versus total knee arthroplasty in patients with isolated patellofemoral osteoarthritis. Am J Orthop (Belle Mead NJ) 2010;39:487-91. [PubMed]
- McKeever DC. Patellar prosthesis. J Bone Joint Surg Am 1955;37-A:1074-84. [Crossref] [PubMed]
- Levitt RL. A long-term evaluation of patellar prostheses. Clin Orthop Relat Res 1973;153-7. [Crossref] [PubMed]
- Vermeulen H, De Doncker E, Watillon M. The Mac Keever patellar prosthesis in femoro-patellar arthrosis. Acta Orthop Belg 1973;39:79-90. [PubMed]
- De Cloedt P, Legaye J, Lokietek W. Femoro-patellar prosthesis. A retrospective study of 45 consecutive cases with a follow-up of 3-12 years. Acta Orthop Belg 1999;65:170-5. [PubMed]
- Hanslik L. Patellofemoral sliding bearing in total knee prosthesis. Preliminary report on the implantation of a modified McKeever endoprosthesis combined with Young's alloarthroplasty. Z Orthop Ihre Grenzgeb 1971;109:435-40. [PubMed]
- Lonner JH. Patellofemoral arthroplasty. Orthopedics 2010;33:653. [PubMed]
- Krajca-Radcliffe JB, Coker TP. Patellofemoral arthroplasty. A 2- to 18-year followup study. Clin Orthop Relat Res 1996;143-51. [Crossref] [PubMed]
- Lonner JH. Patellofemoral arthroplasty: the impact of design on outcomes. Orthop Clin North Am 2008;39:347-54. vi. [Crossref] [PubMed]
- van Jonbergen HP, Poolman RW, van Kampen A. Isolated patellofemoral osteoarthritis. Acta Orthop 2010;81:199-205. [Crossref] [PubMed]
- Borus T, Brilhault J, Confalonieri N, et al. Patellofemoral joint replacement, an evolving concept. Knee 2014;21:S47-50. [Crossref] [PubMed]
- Kazarian GS, Tarity TD, Hansen EN, et al. Significant Functional Improvement at 2 Years After Isolated Patellofemoral Arthroplasty With an Onlay Trochlear Implant, But Low Mental Health Scores Predispose to Dissatisfaction. J Arthroplasty 2016;31:389-94. [Crossref] [PubMed]
- Leadbetter WB, Mont MA. Patellofemoral arthroplasty: a useful option for recalcitrant symptomatic patellofemoral arthritis. Semin Arthro 2009;20:148-60. [Crossref]
- Blazina ME, Fox JM, Del Pizzo W, et al. Patellofemoral replacement. Clin Orthop Relat Res 1979;98-102. [PubMed]
- Kooijman HJ, Driessen AP, van Horn JR. Long-term results of patellofemoral arthroplasty. A report of 56 arthroplasties with 17 years of follow-up. J Bone Joint Surg Br 2003;85:836-40. [PubMed]
- Cartier P, Sanouiller JL, Khefacha A. Long-term results with the first patellofemoral prosthesis. Clin Orthop Relat Res 2005;47-54. [Crossref] [PubMed]
- Tauro B, Ackroyd CE, Newman JH, et al. The Lubinus patellofemoral arthroplasty. A five- to ten-year prospective study. J Bone Joint Surg Br 2001;83:696-701. [Crossref] [PubMed]
- Lonner JH. Patellofemoral arthroplasty: pros, cons, and design considerations. Clin Orthop Relat Res 2004;158-65. [Crossref] [PubMed]
- Laursen JO. High mid-term revision rate after treatment of large, full-thickness cartilage lesions and OA in the patellofemoral joint using a large inlay resurfacing prosthesis: HemiCAP-Wave(R). Knee Surg Sports Traumatol Arthrosc 2016; [Epub ahead of print]. [PubMed]
- Ackroyd CE, Newman JH, Evans R, et al. The Avon patellofemoral arthroplasty: five-year survivorship and functional results. J Bone Joint Surg Br 2007;89:310-5. [Crossref] [PubMed]
- Mont MA, Johnson AJ, Naziri Q, et al. Patellofemoral arthroplasty: 7-year mean follow-up. J Arthroplasty 2012;27:358-61. [Crossref] [PubMed]
- Al-Hadithy N, Patel R, Navadgi B, et al. Mid-term results of the FPV patellofemoral joint replacement. Knee 2014;21:138-41. [Crossref] [PubMed]
- Sarda PK, Shetty A, Maheswaran SS. Medium term results of Avon patellofemoral joint replacement. Indian J Orthop 2011;45:439-44. [Crossref] [PubMed]
- Konan S, Haddad FS. Midterm Outcome of Avon Patellofemoral Arthroplasty for Posttraumatic Unicompartmental Osteoarthritis. J Arthroplasty 2016;31:2657-9. [Crossref] [PubMed]
- Vandenneucker H, Labey L, Vander Sloten J, et al. Isolated patellofemoral arthroplasty reproduces natural patellofemoral joint kinematics when the patella is resurfaced. Knee Surg Sports Traumatol Arthrosc 2016;24:3668-77. [Crossref] [PubMed]
- Cotic M, Forkel P, Imhoff AB. Patellofemoral arthroplasty. Oper Orthop Traumatol 2017;29:40-50. [Crossref] [PubMed]
- Provencher M, Ghodadra NS, Verma NN, et al. Patellofemoral kinematics after limited resurfacing of the trochlea. J Knee Surg 2009;22:310-6. [Crossref] [PubMed]
- Davidson PA, Rivenburgh D. Focal anatomic patellofemoral inlay resurfacing: theoretic basis, surgical technique, and case reports. Orthop Clin North Am 2008;39:337-46. [Crossref] [PubMed]
- Leadbetter WB, Seyler TM, Ragland PS, et al. Indications, contraindications, and pitfalls of patellofemoral arthroplasty. J Bone Joint Surg Am 2006;88:122-37. [PubMed]
- Heyse TJ, El-Zayat BF, De Corte R, et al. Biomechanics of medial unicondylar in combination with patellofemoral knee arthroplasty. Knee 2014;21:S3-9. [Crossref] [PubMed]
- Romagnoli S, Marullo M, Massaro M, et al. Bi-unicompartmental and combined uni plus patellofemoral replacement: indications and surgical technique. Joints 2015;3:42-8. [PubMed]
- Benazzo F, Rossi SM, Ghiara M. Partial knee arthroplasty: patellofemoral arthroplasty and combined unicompartmental and patellofemoral arthroplasty implants--general considerations and indications, technique and clinical experience. Knee 2014;21:S43-6. [Crossref] [PubMed]
- Sabatini L, Schiro M, Atzori F, et al. Patellofemoral Joint Arthroplasty: Our Experience in Isolated Patellofemoral and Bicompartmental Arthritic Knees. Clin Med Insights Arthritis Musculoskelet Disord 2016;9:189-93. [Crossref] [PubMed]
- Laskin RS, van Steijn M. Total knee replacement for patients with patellofemoral arthritis. Clin Orthop Relat Res 1999;89-95. [PubMed]
- Parvizi J, Stuart MJ, Pagnano MW, et al. Total knee arthroplasty in patients with isolated patellofemoral arthritis. Clin Orthop Relat Res 2001;147-52. [Crossref] [PubMed]
- Dalury DF. Total knee replacement for patellofemoral disease. J Knee Surg 2005;18:274-7. [Crossref] [PubMed]
- Meding JB, Wing JT, Keating EM, et al. Total knee arthroplasty for isolated patellofemoral arthritis in younger patients. Clin Orthop Relat Res 2007;78-82. [PubMed]
- van der List JP, Chawla H, Villa JC, et al. Why do patellofemoral arthroplasties fail today? A systematic review. Knee 2017;24:2-8. [Crossref] [PubMed]
- Davies AP. High early revision rate with the FPV patello-femoral unicompartmental arthroplasty. Knee 2013;20:482-4. [Crossref] [PubMed]
- Baker PN, Refaie R, Gregg P, et al. Revision following patello-femoral arthoplasty. Knee Surg Sports Traumatol Arthrosc 2012;20:2047-53. [Crossref] [PubMed]
- Annual Report 2014 Australian Hip and Knee Arthroplasty Register. Australian Orthopaedic Association National Joint Replacement Registry 2014.
- Nicol SG, Loveridge JM, Weale AE, et al. Arthritis progression after patellofemoral joint replacement. Knee 2006;13:290-5. [Crossref] [PubMed]
- Monk AP, van Duren BH, Pandit H, et al. In vivo sagittal plane kinematics of the FPV patellofemoral replacement. Knee Surg Sports Traumatol Arthrosc 2012;20:1104-9. [Crossref] [PubMed]
- Parratte S, Lunebourg A, Ollivier M, et al. Are revisions of patellofemoral arthroplasties more like primary or revision TKAs. Clin Orthop Relat Res 2015;473:213-9. [Crossref] [PubMed]
Cite this article as: Berruto M, Tradati D, Ferrua P, Maione A, Usellini E. Patellofemoral knee arthroplasty. Ann Joint 2017;2:48.


